OUR RESEARCH
Research in the
Kwon Lab for Spinal Cord Injury Repair
The core of our current research program focusses on bridging the gap between scientific discovery and clinical practice with translational research studies that are truly relevant to people with spinal cord injury (SCI). These include studies of how blood pressure management could be optimized to enhance recovery after SCI, how biological responses to injury could be used to identify new targets for treatment and define biomarkers that could be used as biological surrogate outcome measures in a clinical trial of therapeutic for acute SCI. In a collaboration involving several researchers nationwide, we are also dedicated to advance novel implantable technology to study processes occurring inside the spinal cord and bladder, to provide clinicians with real-time physiological information to optimize management of newly injured SCI patients. Moreover, in 2019 we established the International Spinal Cord Injury Biobank (ISCIB) to share valuable SCI tissue specimens with other scientists in an effort to help the international research community move forward faster in the search for new therapies.
See below for an overview of some of our current projects.
CURRENT PROJECTS
Understanding Hemorrhage within the Injured Spinal Cord
This study explores how hemodynamic management and medications to prevent blood clots might influence bleeding within the injured spinal cord. We are conducting experiments to understand the relationship between increasing the blood pressure (to improve spinal cord perfusion) and blood thinners (to prevent blood clots). We are developing novel methods for quantifying hemorrhage using ultrasound, MRI, and histology. Our clinical trial, CHASM, explores this research paradigm in clinical patients who have suffered an SCI (described below).
CHASM
The purpose of the CHASM (Characterizing Hemorrhage in Acute Spinal Cord Injury With MRI) study is to determine whether hemorrhage within the injured spinal cord is influenced by mean arterial pressure (MAP) augmentation with vasopressors and by venous thromboembolism (VTE) prophylaxis with anticoagulants in the first two weeks following a traumatic spinal cord injury (tSCI). SCI patients enrolled in this study will have multiple MRI scans of their spinal cord taken within the first two weeks to characterize how hemorrhage within the cord is progressing.
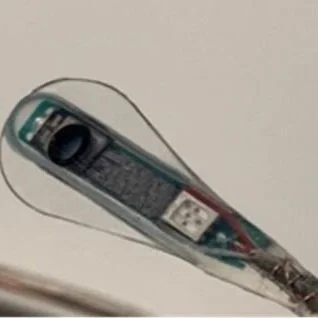
A Novel Biosensor Using Near Infrared Spectroscopy (NIRS)
To understand cord hemodynamics after spinal cord injury, we have developed a biosensor for the spinal cord using Near Infrared Spectroscopy (NIRS), in collaboration with Dr. Babak Shadgan. This sensor non-invasively measures oxygenation & blood flow of the injured spinal cord in real-time. We have evaluated this sensor in our pig model of SCI and demonstrated that it can provide hemodynamic information at the site of injury for upto two weeks. We now plan to evaluate this in a ‘first-in-human’ study of acute SCI patients at Vancouver General Hospital in a clinical trial entitled “OMNI-SCI” for “Optical Monitoring with Near Infrared Spectroscopy for Spinal Cord Injury” (see below). Learn More >
OMNI-SCI
While the management of mean arterial blood pressure (MAP) after acute traumatic spinal cord injury (SCI) is thought to mitigate ischemic, secondary damage, currently no clinical tool exists to measure the impact of MAP on oxygenation and blood flow in the injured spinal cord. In this OMNI-SCI (Optical Monitoring With Near-Infrared Spectroscopy for Spinal Cord Injury) study, we propose to evaluate the safety, feasibility and efficacy of a novel near-infrared spectroscopy (NIRS) system for monitoring oxygenation and hemodynamics in the injured human spinal cord. In addition, this iterative process will allow for further technical refinement with the ultimate goal of optimizing and individualizing hemodynamic management in acute SCI.
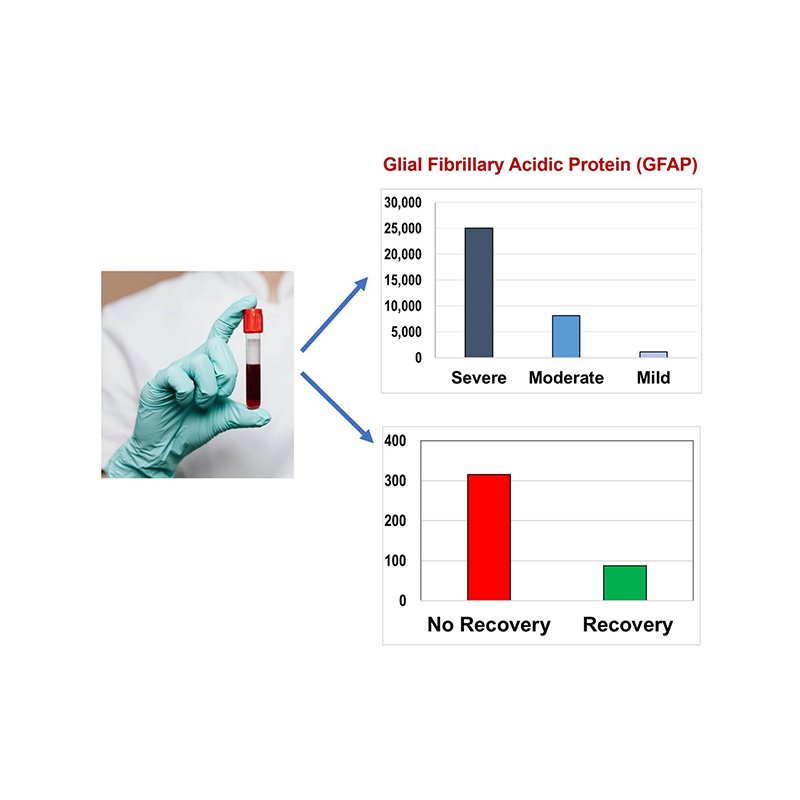
Biomarkers of Spinal Cord Injury
Establishing neuro-chemical biomarkers for acute SCI would help to objectively characterize injury severity and help to prognosticate injury recovery. This is particularly important in the early stages of injury when it is often impossible to perform a standard detailed neurologic examination. We are working with Dr. Cheryl Wellington to establish biomarkers from blood samples of spinal cord injured patients. We have previously done much work in identifying biomarkers in cerebrospinal fluid samples, and while we continue to pursue these studies, we are particularly excited by the potential for biomarkers to be found in blood (which would be much easier to obtain).
ISCIB - International spinal cord injury biobank
The mission of the International Spinal Cord Injury Biobank(ISCIB) is to advance the understanding of the biology of SCI through the study of human specimens. All ISCIB materials are made available to researchers globally, provided that the proposed research has undergone ethical review and is aimed to improve the overall understanding of SCI and the spine. We hope that ISCIB would be accessed by investigators from around the world to address questions about the biology of human SCI that previously may have only been investigated in animal models.
Bridging the Gap with Electronic and Biological Systems
The Bridging the Gap+ study is funded by the United States’ Defense Advanced Research Projects Agency (DARPA). Launched in October 2020, it is a five-year international collaboration, comprising of partners across Canada, the US, and Switzerland. We are developing fully implantable technologies to monitor the status of the injured spinal cord; this information will then be fed into an electrical stimulation system that will control a patient’s blood pressure to optimize delivery of blood and oxygen to the damaged spinal cord tissue. The aim is to see this technology implanted into an acutely injured patient with SCI at Vancouver General Hospital soon.
Monitoring Bladder Pressures After SCI – the Uromonitor
The Uromonitor study in collaboration with Dr. Margot Damaser and Dr. Steve Majerus of the Cleveland Clinic involves the assesses bladder function before and after spinal cord injury using a novel pressure measurement devices. This small, wireless device, known as the “UroMonitor” can be inserted into a patient’s bladder to record its function continuously over multiple fill-empty cycles. Such a device would represent a paradigm shift in the way bladder function is assessed relative to the current “gold-standard” catheter-based method which can often induce pathological symptoms in otherwise ‘healthy’ patients.
Overcoming the Glial Scar – the CSPG Reducing Peptide (CRP)
Dr. Yu Shang Lee and Dr. Ching Li Lin of the Cleveland Clinic has developed a novel peptide (CRP) to degrade the glial scar that forms in the spinal cord after injury. By reducing chondroitin sulfate proteoglycans (CSPGs), a major constituent of the glial scar that inhibits axonal growth and plasticity, functional improvements could be achieved after spinal cord injury (SCI). The role of our lab in this collaborative study is to evaluate the therapeutic effects of CRP in a porcine model of SCI, by examining the anatomical and functional effects of long-term intrathecal CRP treatment.
CASPER
The CASPER (Canadian-American Spinal Cord Perfusion Pressure and Biomarker Study) study is a multi-centre clinical trial examining spinal cord perfusion pressure and SCI biomarkers. The study will help establish clinical guidelines for managing the spinal cord perfusion pressure, which could minimize secondary damage as a result of acute injury and improve recovery. This study will also validate proposed biomarkers for the stratification of injury severity and prediction of neurological recovery. This study is being conducted at eight hospitals across Canada and the US and with an enrolment target of 100 acutely injured patients who have suffered a cervical or thoracic SCI.
SCRIBBLE
The SCRIBBLE (Spinal Cord Injury Blood Biomarker Longitudinal Evaluation) Study is a prospective, single center study at Vancouver General Hospital (VGH) designed to assess blood biomarkers for classifying injury severity and predict neurologic recovery in traumatic spinal cord injured (SCI) patients. This study will also establish the accuracy of point of care devices for SCI blood biomarkers and support the biospecimen collection for the International Spinal Cord Injury Biobank (ISCIB). This study will enroll patients with acute traumatic cervical and thoracic SCI who will have blood samples obtained daily for the first week post-injury. They will then have follow-up neurologic assessments at 6 and 12 months post-injury.
Understanding & Improving Cardiac Function after SCI
In collaboration with Dr. Chris West of ICORD and UBC-Okanagan, we are investigating how acute spinal cord injury in the upper thoracic spine directly impacts the function of the heart, and how this in turn may affect the delivery of blood and oxygen to the injured spinal cord. Understanding these early impact on SCI on heart function is is important because this may represent an critical window of opportunity in the early post-injury period to improve blood and oxygen delivery to the spinal cord, through measures that improve heart function, whilst minimizing the potential for worsened bleeding within the injured spinal cord. This is a very novel approach to the early hemodynamic management of acute SCI patients.
PREVIOUS STUDIES
-

Pro NP
Using Pro-NP developed by Dr. Vinod Labhasetwar from the Cleveland Clinic, a patented antioxidant delivery system of biodegradable nanoparticles to protect and deliver antioxidants, superoxide dismutase and catalases, over a sustained period of time. This study was a platform to formalize the technology.
-

Omics
Using clinical human and research porcine biospecimens, we employ genomic, proteomic, metabolomic, and lipidomic platforms to shed new translational insights into traumatic spinal cord injury (SCI).
-

ACORDA
In this study we evaluated the neuroprotective effects of magnesium chloride within a polyethylene glycol formulation, called "AC105" (Acorda Therapeutics Inc), using a large animal model of SCI.
-

CD11d
The overall goal of this project was to advance the anti-inflammatory neuroprotective CD11d antibody (developed by Eli Lilly and Co.) for use in human SCI clinical trial.
-

Duraplasty
The overall objective of this project was to determine how intraparenchymal spinal cord pressure changes over time after surgical decompression and what effect this has on spinal cord perfusion, oxygenation and downstream metabolic responses in the spinal cord.
-

Triple Monitoring
in this study we evaluated the spatial and temporal dynamics of spinal cord blood flow, tissue oxygenation, pressure, and metabolism in the ‘penumbra surrounding a traumatic SCI.
-

CAMPER
The purpose of CAMPER (Canadian Multicentre CSF Monitoring and Biomarker Study) was to prospectively evaluate spinal cord perfusion pressure (SCPP) in patients with acute spinal cord injuries, to provide scientifically-based guidelines on the management of blood pressure during the acute injury phase.
-

MAP Study
Using our porcine model of SCI, we examined the impact of Mean Arterial Pressure (MAP) augmentation on blood flow, oxygenation, pressure, metabolism, and intraparenchymal hemorrhage within the compressed and decompressed spinal cord.
-

VIbration
Whole-body vibration has been identified as a potential stressor to SCI patients during pre-hospital transportation, particularly for soldiers injured in combat. In collaboration with Dr. Cripton of ICORD, we investigated the consequences of resonance vibration on the injured spinal cord in a porcine model of SCI.
ACADEMIC COLLABORATORS
-

Babak Shadgan
ICORD | University of British Columbia
-

Christopher West
ICORD | University of British Columbia
-

Aaron Phillips
University of Calgary
-

Alex Kavanagh
Vancouver Coastal Health
-

Ching Yi Lin
Cleveland Clinic, USA
-

Gregoire Courtine
EPFL, Switzerland
-

Jocelyne Bloch
Lausanne University Hospital | EPFL, Switzerland
-

John 'Kip' Kramer
ICORD | University of British Columbia
-

Margot Damaser
Cleveland Clinic, USA
-

Yu Shang Lee
Cleveland Clinic, USA
-

Karen Moxon
University of California at Davis, USA
-

Mark Tuszynski
University of California at Davis , USA
-

Peter Cripton
ICORD | University of British Columbia
-

Wolfram Tetzlaff
ICORD | University of British Columbia
-

Leonard Foster
University of British Columbia
-

Sean Christie
Dalhousie University
-

Jean Marc Mac-Thiong
University of Montreal
-

Liang Li
University of Alberta
-

Kendall van Keuren Jensen
Translational Genomics Institute, USA
-

Bruce McManus
PROOF Center | University of British Columbia
-

Don Griesdale
University of British Columbia
-

Mypinder Sekhon
University of British Columbia
-

Andrew Macnab
University of British Columbia
-

Guy Dumont
University of British Columbia
-

Michael Fehlings
Univ of Toronto
-

Vinod Labhasetwar
Cleveland Clinic, USA
-

Sanjay Dhall
University of California at Los Angeles, USA
-

Lynn Stothers
University of British Columbia
-

Corree Laule
ICORD | University of British Columbia
-

Tom Oxland
ICORD | University of British Columbia
-

Wayne Moore
ICORD | University of British Columbia
-

Piotr Kozlowski
University of British Columbia
-

Ona Bloom
The Feinstein Institute for Medical Research, USA
-

Christoph Borchers
University of Victoria
-

Cheryl Wellington
University of British Columbia
-

Corey Nislow
University of British Columbia
-

Patricia Jill Ward
Emory University, USA
-

Veronica Hirsh-Reinshagen
University of British Columbia
-

Maxwell Boakye
University of Louisville, USA
-

Charles Hubscher
University of Louisville, USA
-

Jamie Wilson
University of Nebraska, USA
-

Christian Ricks
Univ. of New Mexico, USA
-

Raju Heran
University of British Columbia
-

Nathan Evanview
University of Calgary
-

Anthony DiGiorgio
University of California - San Francisco, USA
-

David Okonkwo
University of Pittsburgh, USA
-

Jeff Wilson
University of Toronto
We are grateful to be supported by
-

US Department of Defence
-

US Defense Advanced Research Projects Agency
-

Craig H Neilsen Foundation
-

Canadian Institutes of Health Research
-

Wings for Life Foundation
-

Brain Canada
-

MITACS
-

Michael Smith Foundation for Health Research
-

Praxis Spinal Cord Institute
-

University of British Columbia-Faculty of Medicine
-

New Frontiers in Research Fund
-

Rick Hansen Foundation
-

VGH+UBC Foundation
-

Djavad Mowafaghian Foundation
-
-
-